From time to time proteins misfold. When that comes about in the human brain, the pileup of misfolded
proteins can direct to neurodegenerative diseases like Alzheimer’s, Parkinson’s and
ALS.
Proteins do not misbehave and misfold out of the blue. There is a sensitive ecosystem
of biochemical interactions and environments that normally allow them twist, unfold,
refold and do their jobs as they are meant to.
However, as researchers from Michigan Technological University check out in an posting
published in ACS Chemical Neuroscience, even a compact adjust might bring about extensive-phrase outcomes.
For amyloid beta peptides — considered a main hallmark of Alzheimer’s sickness — a
widespread chemical modification at a specific area on the molecule has a butterfly
outcome that sales opportunities to protein misfolding, aggregation and mobile toxicity.
Acetylation and Amyloid Beta Toxicity
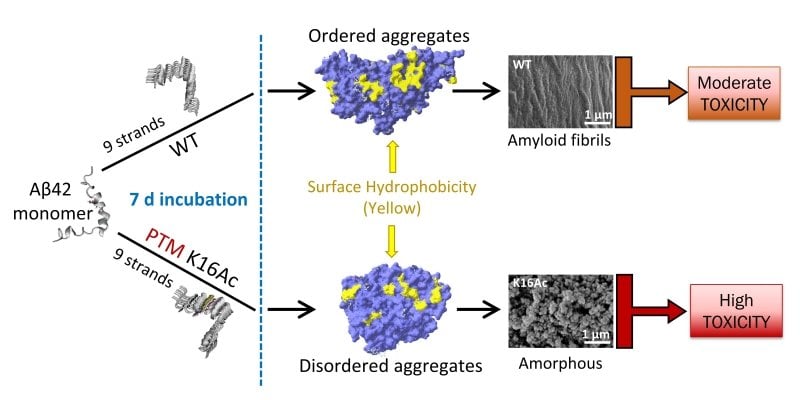
Ashutosh Tiwari, associate professor of chemistry at Michigan Tech, describes that misfolded amyloid beta proteins have a tendency to pile up and
kind aggregates, which can kind stringy fibrils or balled-up amorphous shapes.
To understand what results in the distinctive shapes and to assess their toxicity, Tiwari’s
team looked at acetylation.


misfolded proteins producing extensive, stringy shapes. Credit score: Ashutosh Tiwari
Acetylation is a person of the most widespread chemical modifications proteins undergo, but
a person of the least researched in phrases of how it impacts amyloid beta toxicity. On amyloid
beta proteins, acetylation can arise at two web pages: lysine sixteen and lysine 28.
The team observed that acetylation at lysine sixteen led to the disordered aggregates that
shaped sticky but adaptable amorphous buildings and showed higher ranges of toxicity.
They also observed the aggregates showed higher free of charge radical development.
“No a person has done a systematic analyze to display if you acetylate amyloid beta it modifications
how the mixture seems to be, then it modifications its biophysical attributes and hence toxicity,”
Tiwari said. “What we’re stating is that the shape, stickiness and flexibility of the
aggregated protein composition can participate in a vital position in the mobile toxicity and might
also affect the system of toxicity.”
Aggregates and Alzheimer’s
In Alzheimer’s, these aggregates accumulate in the aspect of the brain that impacts
memory. It’s a sickness that the Alzheimer’s Corporation studies is the sixth foremost
bring about of loss of life in the US and will price tag the country about $305 billion in 2020. Tiwari
claims what we truly need to have to understand about the sickness is that there is no one
bring about, no one induce, and almost certainly no silver bullet mainly because of the chemistry involved.
Grants and Funding
Investigate Excellence Fund, Michigan Technological University, Linda J. Horton Laboratory
Investigate Fund and Protein Misfolding Diseases Investigate Fund, Nationwide Institutes of
Health NIH R15 HL129213
“This is how a delicate adjust on a one placement can affect a entire protein’s aggregation,”
Tiwari said, adding that the outcome of acetylation on tau, one more protein aggregation,
has been far a lot more studied than amyloid beta. Also, a lot of researchers continue to feel a
misfolded protein has to search a sure way to turn into problematic, and that other
misfolded forms are fewer of an challenge.
Tiwari agrees some of the proteins’ modifications are delicate, and compares discerning the
distinctions and their effects to snow tires. Snow tires have deeper treads and a a lot more
adaptable content to cope with wintertime roadways, but it’s difficult to level out people capabilities
at highway speeds. Like distinctive forms of tires, protein shapes can look indistinguishable
at a length.
“This is not some thing that can be viewed from afar — it’s a contact-and-come to feel assets,”
Tiwari said. “We have to interrogate these attributes. We have to search at these buildings
a lot more deeply from each morphology and biophysical views.”
When we do, we might far better understand the complexity of the misfolded proteins and
amyloid beta toxicity that can bring about neurodegenerative diseases like Alzheimer’s.
modifications in protein folding chemistry can bring about distinctions in shape and toxicity.
Credit score: Ashutosh Tiwari
Michigan Technological University is a public research college, household to a lot more than
seven,000 learners from 54 international locations. Established in 1885, the University features a lot more than
120 undergraduate and graduate degree plans in science and engineering, engineering,
forestry, business enterprise and economics, overall health professions, humanities, mathematics, and
social sciences. Our campus in Michigan’s Higher Peninsula overlooks the Keweenaw Waterway
and is just a number of miles from Lake Remarkable.




